Humoral immunity against infections depends on the germinal center (GC) reaction occurring in the B cell follicles of lymphoid tissues, e.g. lymph nodes. Upon T cell-dependent activation, naïve B cells form GCs, wherein they undergo massive proliferation and somatic hypermutation of their immunoglobulin loci, to eventually form antibody-secreting plasma cells. The process of GC reaction is further controlled by epigenetic switches. A major impediment to detailed mechanistic studies of normal and transformed GC B cells, as well as the ability to generate high affinity, antibody secreting cells in vivo, is the lack of experimental systems that sufficiently recapitulate the biology of this complex biological system. Overcoming this limitation requires the ability to manipulate and generate such cells in a tractable system. Our work has led to the development of a first immune tissue in a dish that recapitulates selective aspect of lymph nodes and germinal centers. See our Nature Communications, Nature Protocols, Biomaterials, and ACS Biomaterials Science and Engineering papers. Our interest areas include:
(1) Understanding of factors that control B and T cell immune response against threats;
(2) Rapid development and translation of a large number of activated B cells capable of producing disease-specific antibodies;
(3) Rational development and evaluation of immunotherapeutics and vaccines against HIV, Zika, and other infectious diseases, and
(4) Understanding matrix immunology and B cell receptor signaling
This research was highlighted as Top 100 discoveries of 2015 by the Discover Magazine and a recipient of 2015 Biomaterials Outstanding Paper Award from Elsevier.
See Cornell Chronicle: http://news.cornell.edu/stories/2015/06/engineers-synthetic-immune-organ-produces-antibodies
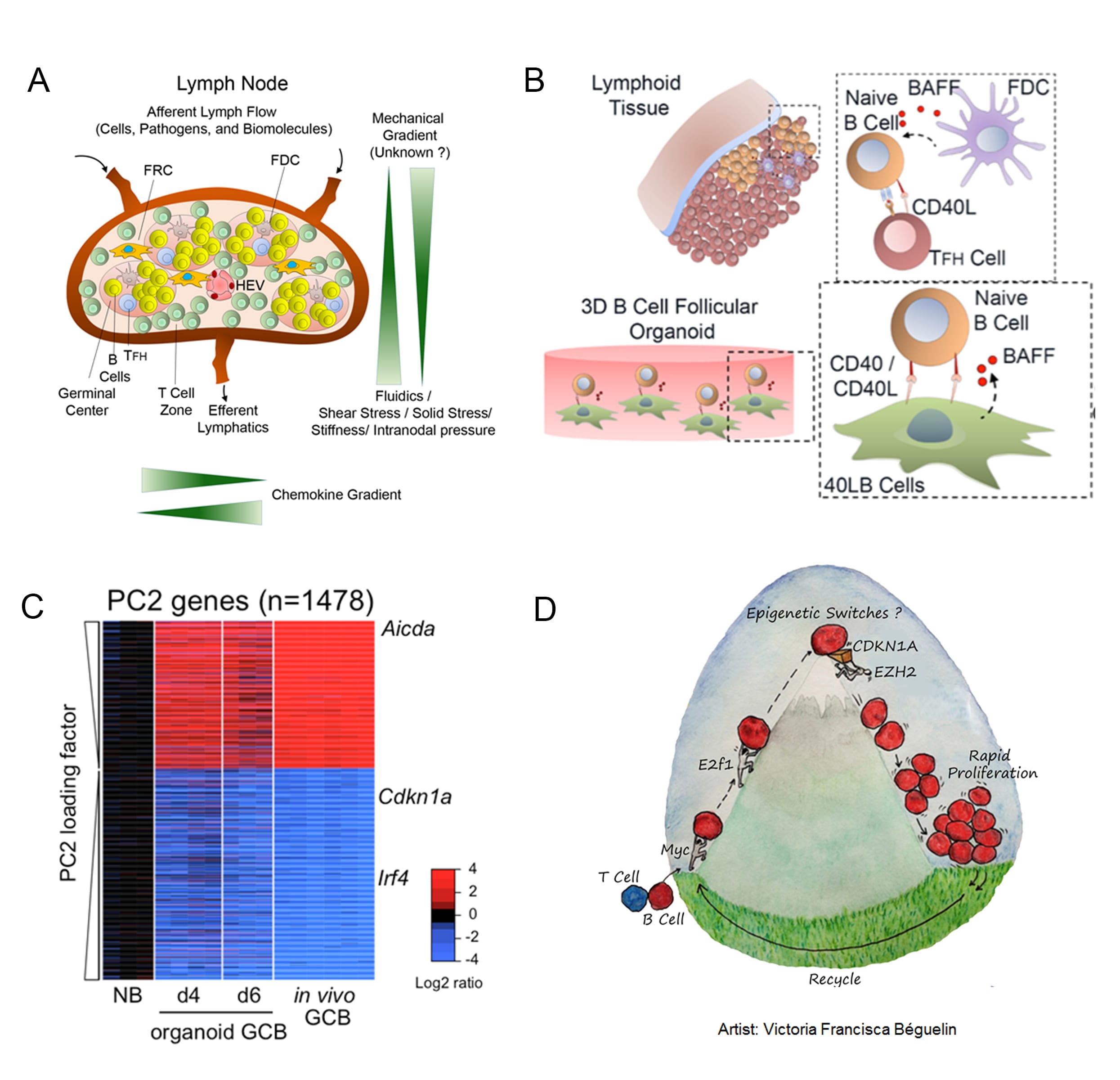
Figure: Schematic with cellular, biophysical, and biochemical components of the lymph node (Panel A). Ex vivo 3D bioengineered immune organoids induce rapid differentiation of naive B cells into germinal center-like phenotype within 4 days and at ~ 100-fold faster rate than conventional ex vivo immunology approaches (Panel B). The whole transcriptome is comparable to immunized mice (Panel C) and enabled a recent discovery of epigenetic modulator of the germinal center process (Panel D; Nature Communications, 2017)