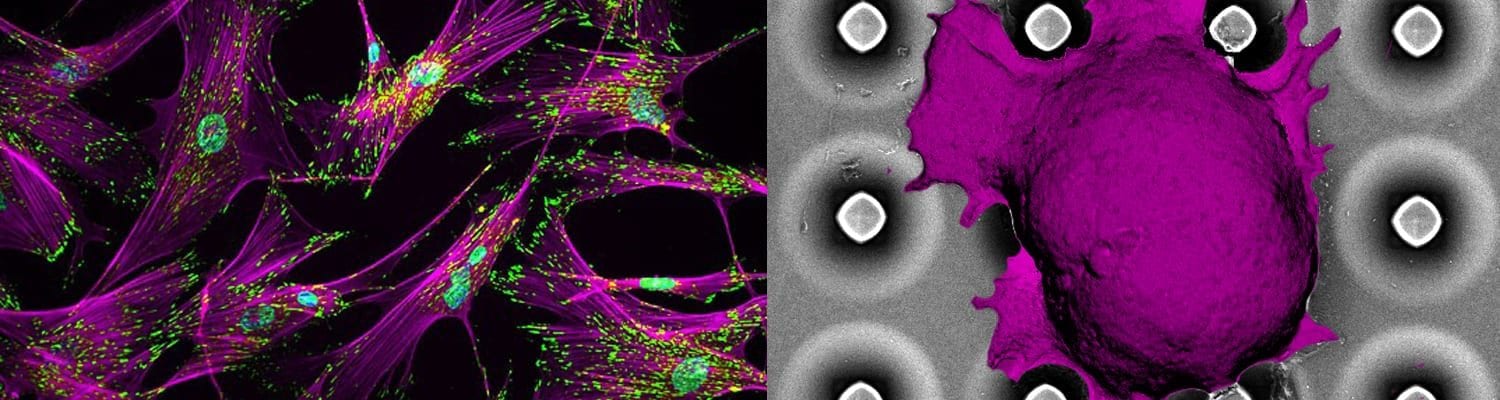
Tumor growth and survival are maintained by cellular and molecular mechanisms linked either to aberrant mutations or to normal proteins hijacked by tumors to their advantage. Targeting hallmark proteins and pathways hold the promise to improve efficacy and decrease the toxicity of anticancer treatments. However, even targeted therapies are unlikely to be curative unless their therapeutic efficacy is evaluated under appropriate tumor microenvironment that represents complexity, heterogeneity, feedback mechanisms, and propensity to develop resistance. Our research focus has been on understanding the role of lymphoid tumor microenvironment in B and T cell lymphomas and developing innovating biomaterials-based platform technologies to determine tumor heterogeneity and causes of drug resistance in lymphoma patients. We apply concepts of tissue mechanics, tissue bio-adhesivity, and fluid mechanics integrated with cancer cell biology to engineer platform technologies that enable fundamental discoveries in cancers biology and rational translation of therapeutics. Our recent findings with collaborators at Weill Cornell Medical College, published in Blood, demonstrated the role of integrin signaling in patient-derived T cell lymphoma survival and progression, in vitro and vivo. In a parallel study published in Biomaterials, we have provided evidence that integrins are critical for the growth, clustering, BCR activity, and chemo-resistance of activated B cell-like Diffuse Large B cell lymphoma (ABC-DLBCL), which are among the most chemo-resistant lymphomas. These biological discoveries led to the development of the first 3D lymphoma organoids (hydrogels) that presented lymphoma-specific integrin ligands to ABC-DLBCL and induced enhanced proliferation, cell signaling, and drug-resistant.
Cornell Chronicle: http://www.news.cornell.edu/stories/2015/10/3-d-organoids-allow-tests-lymphoma-treatments
Cornell Chronicle: Immune-engineered device targets chemo-resistant lymphoma
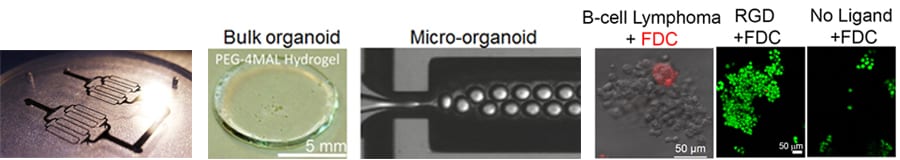
Figure: B cell lymphomas grow in bulk and micro organoids as clusters, similar to those in patients. The green fluorescent areas represent lymphoma cells